Recap from our last update: We narrowed our focus from child mortality to pneumonia (15% of all <5 deaths globally), and started looking at general ways to approach ending pneumonia.
Over this last week, we shifted our focus from oxygen therapy to vaccines + scoped our location down to Uttar Pradesh or Bihar in India.
Here’s what you’ll learn from this update:
Why prevention > treatment when it comes to pneumonia
Why we’ve chosen Uttar Pradesh/ Bihar
Why storage/ distribution is the biggest bottleneck in India’s cold chain
Our next steps
See a video summary of this newsletter: here
Treating vs Preventing Pneumonia
Last update, we were looking at oxygen therapy for hypoxemia, which occurs in 13% of all children with pneumonia. Our thought process for tackling oxygen therapy was as follows:
Oxygen therapy impacts severe pneumonia cases: Children with severe pneumonia + hypoxemia are five times as likely to die, and improved oxygen delivery systems could reduce childhood pneumonia related mortality by at least 35%.
Oxygen therapy is a widely used treatment: O2 is also used for the treatment of many other common respiratory (tuberculosis, COPD, COVID), non-respiratory diseases (malaria, asphyxia, eclampsia) + other post-operative complications.
The crux of our decision was that improving oxygen = not only helping with pneumonia, but also with the treatment of many other diseases.
As we learned more about oxygen therapy and the gaps within the pipeline, we decided to pivot away from the treatment side of things, and towards prevention. Here’s why:
In the best case scenario, if we were able to increase access to oxygen therapy, children are still going to get pneumonia. Even if you can help treat the 13% of children with pneumonia + hypoxemia, children are still getting pneumonia + themselves and their families have to deal with the physical, emotional, and financial impacts of disease.
Moreover, simply providing access to oxygen isn’t the sole solution - depending on the source (cylinder, concentrator, etc), you need constant electricity, a solid supply chain for maintenance and repair + sourcing of spare parts, and the O2 therapy needs to be accompanied with other treatments (e.g. antibiotics) to effectively tackle the disease. Depending on the country, sourcing the many requirements can be difficult.
Our big vision is to make the world exponentially better. And although treatments like antibiotics and oxygen therapy (among other things) are incredibly essential, they aren’t proactive measures and are still accepting a world where children are getting pneumonia.
Our New Focus: Using vaccines to prevent pneumonia from occurring
The PCV vaccine (pneumococcal conjugate) is the best way to prevent childhood pneumonia - the PCV13 is 86% effective and dramatically reduces the number of <5 deaths. Despite this, over 50% of children around the world are not immunized with the PCV vaccine.
Our question is - how can we get more children vaccinated with the PCV vaccine?
Although there are common issues within the immunization process, problems can vary on a location-specific basis.
Scoping an ideal location - our process:
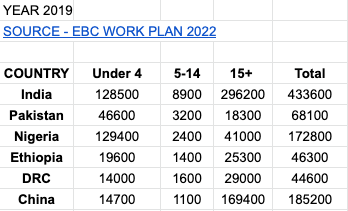
80% of all <5 deaths occur in 6 countries - China, India, Pakistan, Nigeria, Ethiopia and the Democratic Republic of the Congo. Using data from Every Breath Counts Coalition’s 2022 Work Plan, we mapped out the number of pneumonia deaths + age range:
When graphing the under 4 deaths, India and Nigeria cumulatively account for 73% of these deaths. Between India and Nigeria, we decided to focus on India. Our thought process:
In India, Uttar Pradesh (UP) and Bihar have the highest estimated number of pneumonia cases - 12.4 million and 7.3 million respectively.
Mapping out the Pneumonia Cold Chain
Sourcing Vaccines
India has secured 69 million doses of pneumonia vaccines: 60 million doses of PCV13 through the Gavi Pneumoccocal Advance Market Commitment mechanism, and 9 million through the Serum Institute, who developed India’s first native pneumonia vaccine called Pneumosil. We estimate that this is sufficient for 23 million children.
Link to spreadsheet
The Major Challenge in India: Storage and Distribution
In India, the vaccine typically goes from the factory to GMSDs (government medical store depots) to state vaccine stores, after which they are taken to regional/ district/ sub-district locations for administration.
The cold chain in India however, travels from factories to CFAs (carrying and forwarding agents) to distributors. Depending on the particular region and its population (density, proximity, and urban/rural), the distributor may not deliver vaccines to the end consumer (at the sub-district locations) - this is especially prominent in rural areas.
The big idea: last mile delivery is complicated and not always accounted for. This often leads to under the table deals with the aim to ‘get the job done.’ This is dangerous as this risks the vaccines to temperature damage, which can reduce vaccine potency.
When evaluating potency at other parts of the supply chain, appearance is not a reliable indicator → You can’t tell if the vaccine is inactive because of temperature damage.
During transportation, vaccines are stored on truck refrigeration units with continuous power supplies to ensure the vaccines are kept cool. This can become expensive -
Diesel as a source of electricity can be very expensive for smaller packages and shipments
Because of India’s humid weather, multi-temperature fleets are often required ⇒ becomes expensive
Where are we right now? + Next Steps:
We’ve identified one of the most pressing parts of the vaccine administration process - actually transporting vaccines to the correct locations without facing temperature damage. Our next steps are to focus on problem hypotheses to identify the specific, niche problem that is impact driven, but also economically incentivized.
Another thing we’re planning on focusing on is understanding the cold chain + immunization landscape in India, but also other locations with effective cold chains. The process briefly outlined above isn’t specific only to pneumonia vaccines - our plan is to look at COVID vaccines + food products to find case studies/ transferrable strategies.
Shoutouts:
Roger + Maire: thank you so much for providing us with valuable insights and taking the time to help us validate our assumptions! We’re very excited and we’re looking forward to more collaboration in the future :)
Our Asks:
We’re currently looking for connections to anyone working in vaccine distribution, cold storage, pneumonia vaccines, healthcare in developing countries, or pneumonia organizations in general.
For any questions or to collaborate, email at richapandya273@gmail.com or m.roopkalsi5@gmail.com.
See you soon!
Richa & Manroop ❤️🔥